Your gift is 100% tax deductible.
Gastrointestinal Stromal Tumor (GIST)
If you have a gastrointestinal stromal tumor (GIST) or are close to someone who does, knowing what to expect may be helpful. Here you can find out all about GISTs, including risk factors, symptoms, and how they are diagnosed and treated.
About gastrointestinal stromal tumors
GISTs are a rare type of cancer. GIST tumors are sarcomas. Sarcomas are cancers that develop from cells that normally make up connective tissue in the body, such as muscles, fat, bones, the lining of joints, or blood vessels.
GISTs start in early forms of cells called the interstitial cells of Cajal (ICCs). ICCs are in the wall of the gastrointestinal (GI) or digestive tract. They have been called “pacemakers” because they normally signal the muscles in the GI tract to contract to move food and liquid along.
More than half of GISTs grow in the wall of the stomach. Most of the others start in the small intestine, but GISTs can start anywhere along the GI tract.
A small number of GISTs start outside the GI tract in nearby areas such as the omentum (a layer of fatty tissue that hangs over the organs in the abdomen) or the peritoneum (the thin lining over the organs and walls inside the abdomen).
Some GISTs seem to be much more likely than others to come back (recur) after surgery, grow into other areas, or spread to other parts of the body. Factors that may indicate a GIST tumor is more likely to recur, grow, or spread quickly include:
- The size of the tumor
- Where it's located in the GI tract
- How fast the tumor cells are dividing (its mitotic rate)
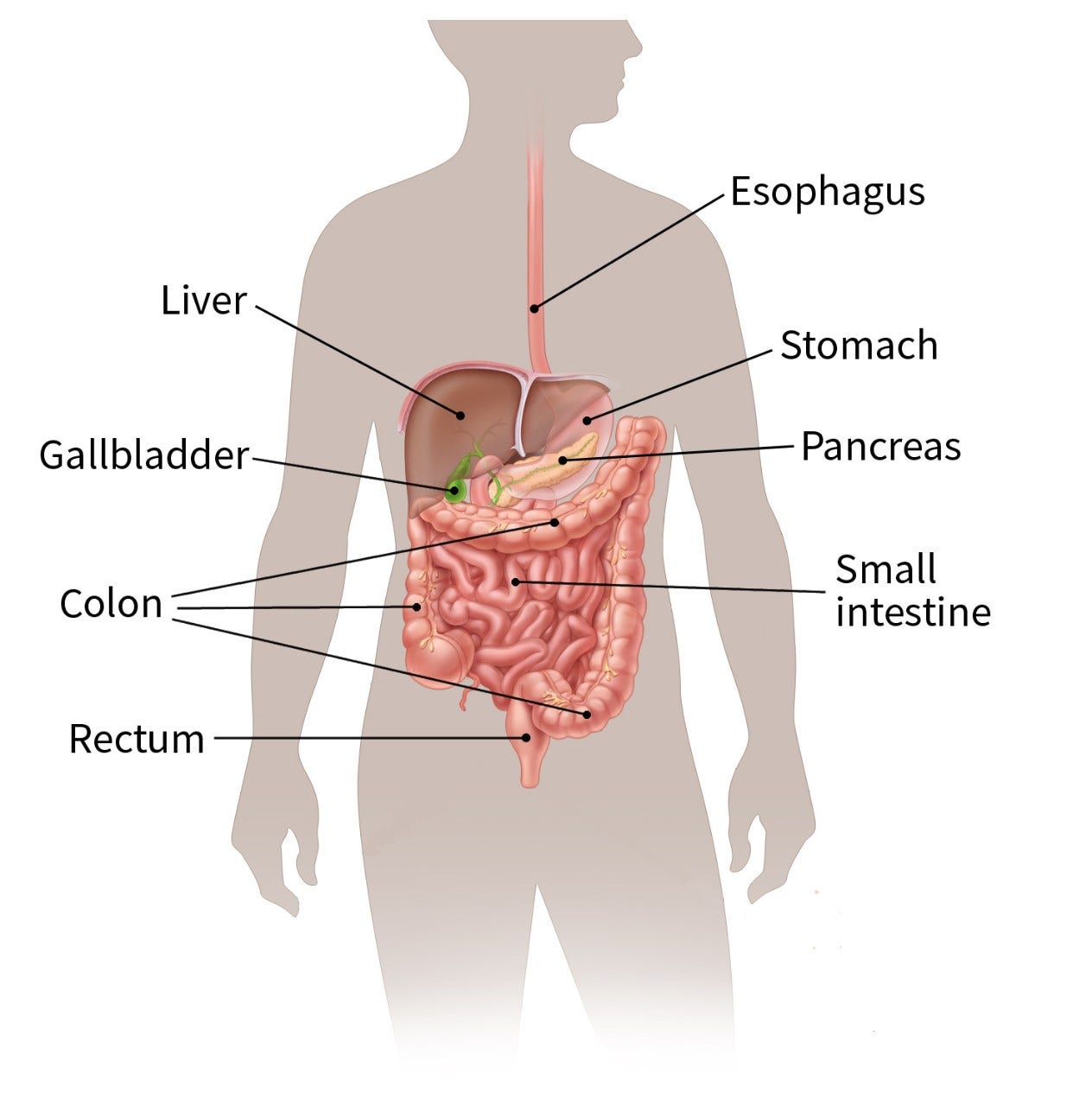
The gastrointestinal (GI) tract
The GI tract is a long tube connecting the mouth to the anus, which processes food for energy and removes solid waste.
After food is chewed and swallowed, it goes through the esophagus, a tube that carries food down the throat and chest to the stomach. The esophagus joins the stomach just below the lungs.
The stomach is a sac-like organ that helps the digestive process by mixing the food with gastric juices. The food and gastric juices are then emptied into the small intestine. The small intestine, which is about 20 feet long, continues breaking down the food and absorbs most of the nutrients into the bloodstream.
The small intestine joins the large intestine, the first part of which is the colon, a muscular tube about 5 feet long. The colon absorbs water and minerals from the food. The waste left after this process (stool) goes into the rectum, where it is stored until it passes out of the body through the anus.
The interstitial cells of Cajal (ICCs) are present throughout the GI tract and help move food and liquid. GISTs develop from these cells.
Other cancers of the GI tract
GISTs are rare tumors that start from ICCs, but there are several other types of cancers that start in the GI tract. Doctors need to find out if a tumor in the GI tract is a GIST, some other type of cancer, or a non-cancerous condition to decide on the best treatment.
Common GI tract cancers
The most common GI tract cancers are:
- Adenocarcinomas, which start in the gland cells that line almost all of the GI tract. These include most esophagus cancers, stomach cancers, and colon and rectum cancers.
- Squamous cell carcinomas, which start in squamous cells. These are flat cells that line some parts of the GI tract, like the upper part of the esophagus and the end of the anus.
More information about these more common cancers of the digestive system can be found here:
Rare GI tract cancers
GI neuroendocrine tumors (NETs): These tumors, also called GI carcinoid tumors, form from neuroendocrine cells in the GI tract. These cells have some features in common with nerve cells but also have other features in common with hormone-producing (endocrine) cells.
Other rare types of cancer in the GI tract include different types of soft tissue sarcomas, such as:
- Leiomyosarcomas: cancers of smooth muscle cells
- Angiosarcomas: cancers of blood vessel cells
- Malignant peripheral nerve sheath tumors (MPNSTs): cancers of cells that support and protect nerves
- Written by
- References

Developed by the American Cancer Society medical and editorial content team with medical review and contribution by the American Society of Clinical Oncology (ASCO).
National Cancer Institute. Gastrointestinal Stromal Tumors Treatment. Accessed at https://www.cancer.gov/types/soft-tissue-sarcoma/hp/gist-treatment-pdq on December 4, 2025.
National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology: Gastrointestinal Stromal Tumors Version 1.2025 – April 17, 2025. Accessed at https://www.nccn.org/professionals/physician_gls/pdf/gist.pdf on December 4, 2025.
Raut CP, Duensing A, Keedy VL. Clinical presentation, diagnosis, and prognosis of gastrointestinal stromal tumors. UpToDate. 2025. Accessed at https://www.uptodate.com/contents/clinical-presentation-diagnosis-and-prognosis-of-gastrointestinal-stromal-tumors on December 4, 2025.
Van Tine BA. Chapter 90: Sarcomas of Soft Tissue. In: Niederhuber JE, Armitage JO, Dorshow JH, Kastan MB, Tepper JE, eds. Abeloff’s Clinical Oncology. 6th ed. Philadelphia, Pa. Elsevier: 2019.
Last Revised: March 10, 2026
American Cancer Society medical information is copyrighted material. For reprint requests, please see our Content Usage Policy.



