ACS Research News
The State of Survivorship: Prevalence, Projections, & Gaps in Care
Published on: July 9, 2025
A new American Cancer Society (ACS) study reports on cancer survivorship—and where treatment equity still falls short.
Healthy Lifestyle Improves Survival from Obesity-Related Cancers
Published on: June 24, 2025
CPS-II Nutrition data shows that people with obesity-linked cancers lower their risk of death by sticking with the ACS Nutrition & Physical Activity Guideline.
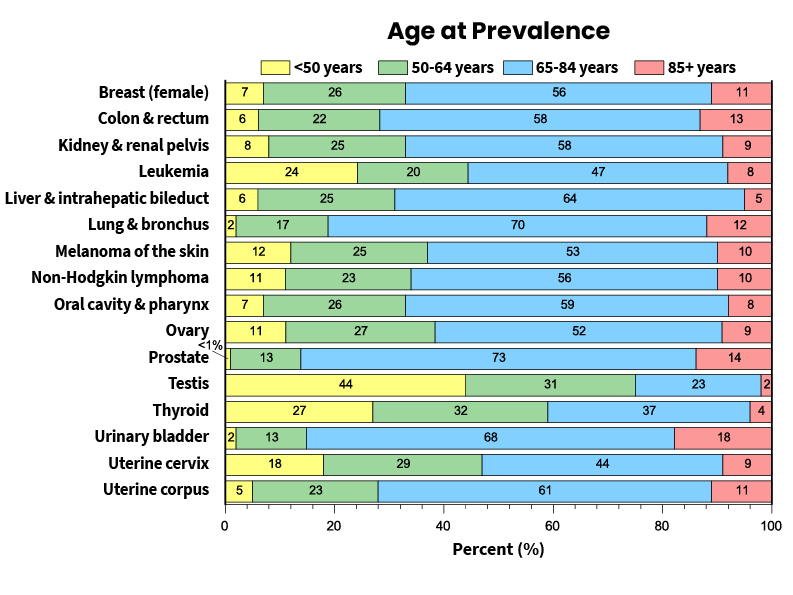
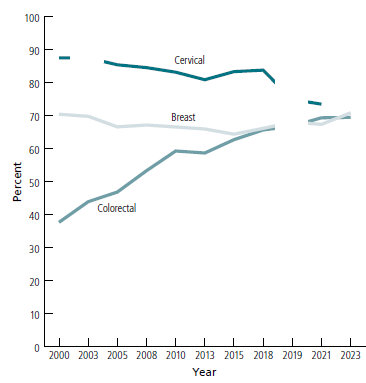
Not Enough Females Are Being Screened for Cervical Cancer
Published on: April 25, 2025
Cancer Prevention & Early Detection report says cervical cancer prevention needs urgent attention. Good news: Fewer people smoke than ever before.