Your gift is 100% tax deductible.
Esophageal Cancer
If you have esophageal (esophagus) cancer or are close to someone who does, knowing what to expect can help you cope. Here you can find out all about esophageal cancer, including risk factors, symptoms, how it is found, and how it is treated.
About esophageal cancer
Esophageal cancer (also called esophagus cancer or cancer of the esophagus) starts when cells in the lining of the esophagus begin to grow out of control.
Esophageal cancer can start anywhere along the esophagus. It starts in the inner layer of the esophagus wall (see below), and it then grows outward through the other layers.
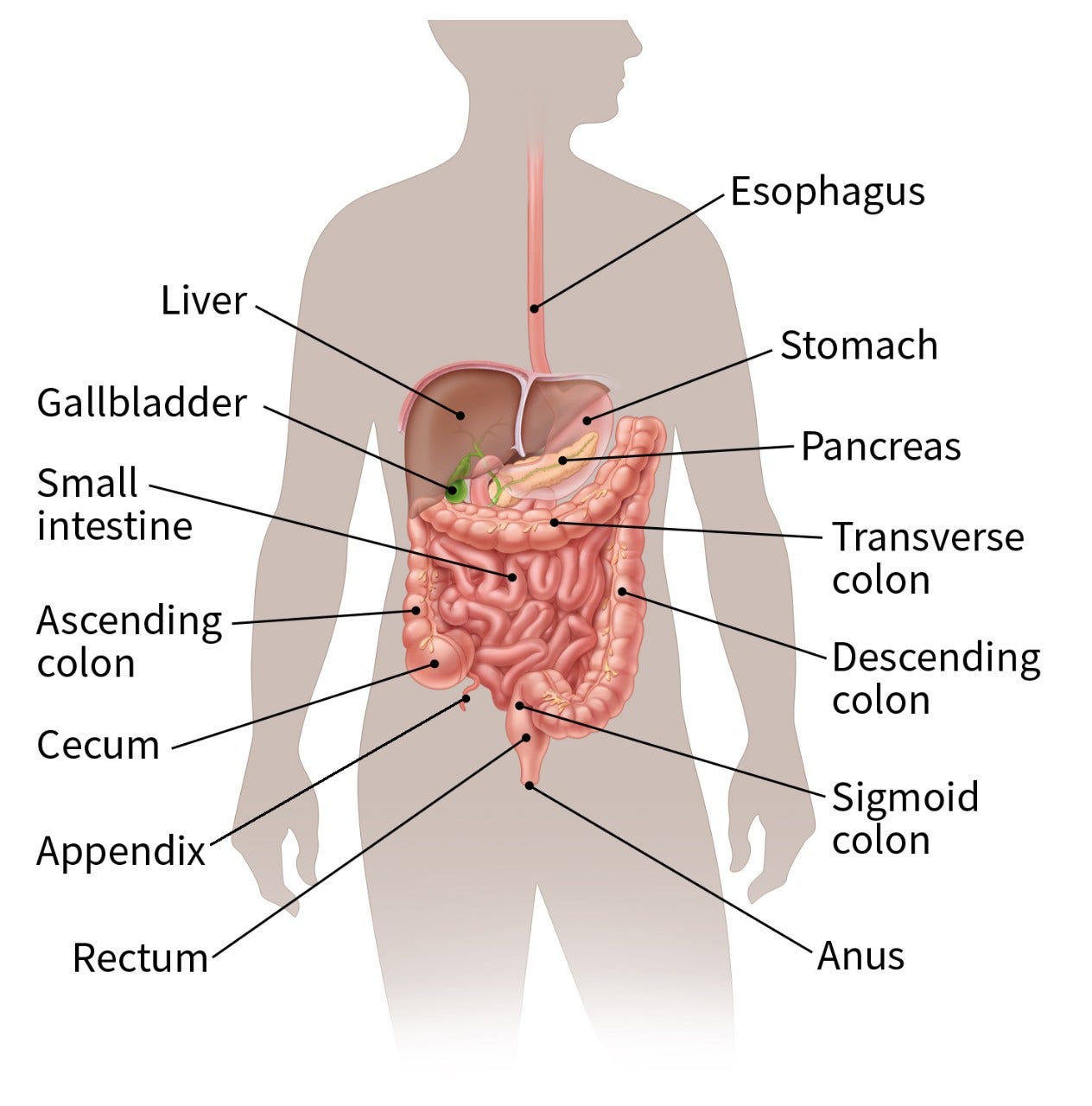
Understanding the esophagus
To understand esophageal cancer, it helps to know about the normal structure and function of the esophagus.
The esophagus is a hollow, muscular tube that allows food and liquids to pass from the throat to the stomach. It lies behind the trachea (windpipe) and in front of the spine.
Parts of the esophagus
At the opening of the upper esophagus is a special ring of muscle (called the upper esophageal sphincter) that opens the esophagus when it senses food or liquid coming toward it.
When you swallow, food and liquids travel through the inside of the esophagus (called the lumen) to reach the stomach.
The lower part of the esophagus that connects to the stomach is called the gastroesophageal junction (GEJ), also known as the esophagogastric junction (EGJ).
A special ring of muscle near the junction, called the lower esophageal sphincter, controls the movement of food from the esophagus into the stomach. Between meals, it stays closed to keep the stomach’s acid and digestive juices from moving up into the esophagus.
© American Society for Clinical Oncology. Used with permission.
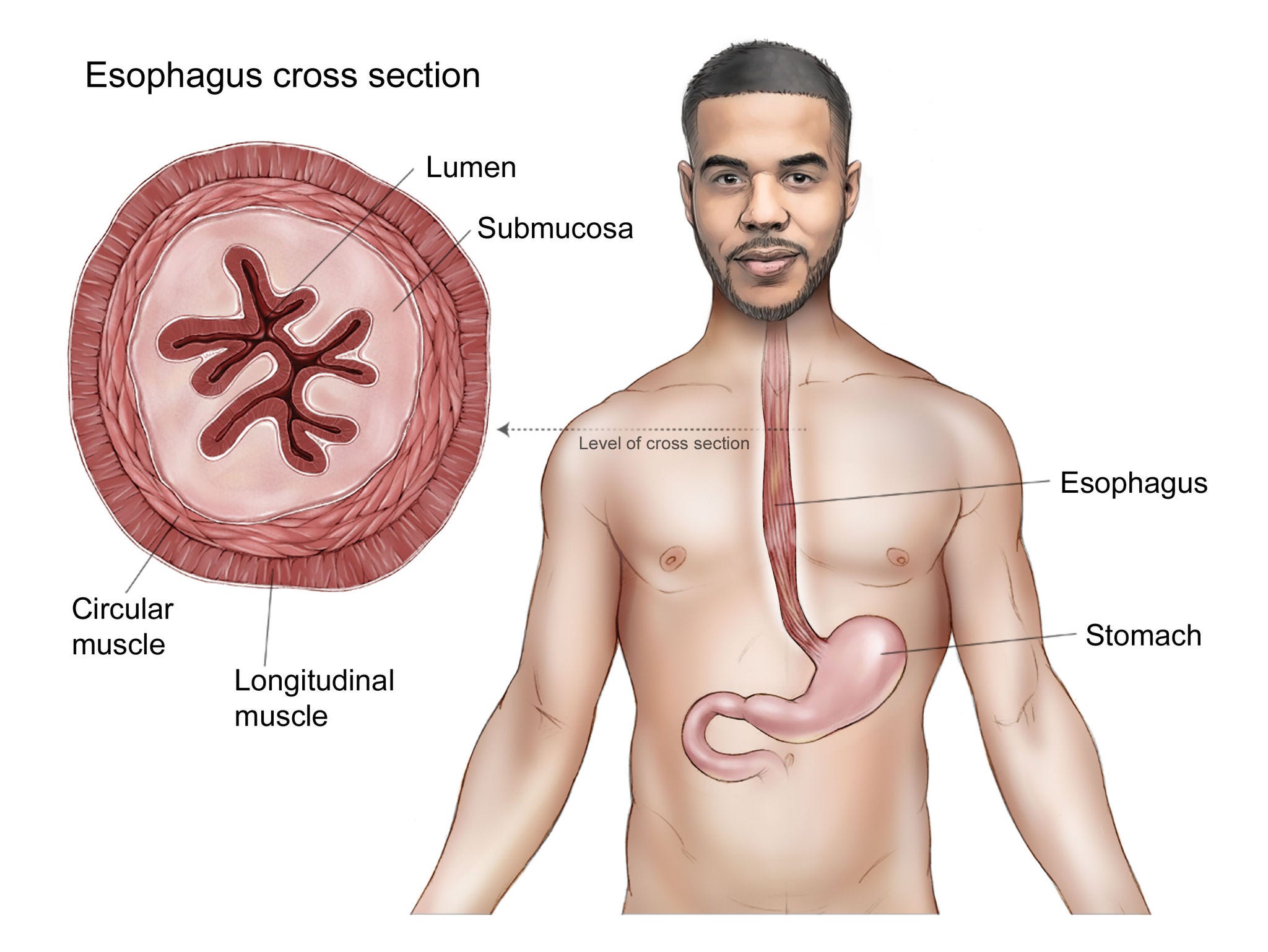
The esophagus wall
The wall of the esophagus has several layers:
Mucosa: This layer lines the inside of the esophagus. It has 3 parts:
- Epithelium: The innermost lining of the esophagus. It is made up mostly of flat, thin cells called squamous cells. This layer is where most esophageal cancers start.
- Lamina propria: A thin layer of connective tissue right under the epithelium
- Muscularis mucosa: A very thin layer of muscle under the lamina propria
Submucosa: This is a layer of connective tissue just below the mucosa that contains blood vessels and nerves. In some parts of the esophagus, this layer also includes glands that secrete mucus.
Muscularis propria: This is a thick layer of muscle under the submucosa. It contracts in a coordinated way to push food down the esophagus from the throat to the stomach.
Adventitia: This is the outermost layer of the esophagus. It is made up of connective tissue.
Types of esophageal cancer
There are 2 main types of esophageal cancer, based on the type of cell it starts in.
Squamous cell carcinoma
The inner layer of the esophagus (the mucosa, see above) is normally lined with squamous cells. Cancer starting in these cells is called squamous cell carcinoma. This type of cancer can occur anywhere along the esophagus, but it is most common in the middle part of the esophagus.
Squamous cell carcinoma used to be the most common type of esophageal cancer in the United States. This has changed over time. Now, it makes up less than 1 in 3 esophageal cancers in this country. However, it’s still the most common type of esophageal cancer in many parts of the world.
Adenocarcinoma
Cancers that start in gland cells (cells that make mucus) are called adenocarcinomas. Adenocarcinomas are most often found in the lower third of the esophagus. This is now the most common type of esophageal cancer in the US.
These cancers are often linked to conditions such as Barrett's esophagus, in which gland cells have replaced the squamous cells in the mucosa in the lower part of the esophagus.
Gastroesophageal junction (GEJ) tumors: Adenocarcinomas that start at the area where the esophagus joins the stomach (the GEJ, see above), including the first couple of inches of the stomach, tend to behave like esophageal cancers and are treated like them as well.
Rare cancers in the esophagus
Other types of cancer can also start in the esophagus, including:
These cancers are rare and are not discussed further here.
Quick guides
- Written by
- References

Developed by the American Cancer Society medical and editorial content team with medical review and contribution by the American Society of Clinical Oncology (ASCO).
Gibson MK. Epidemiology and risk factors for esophageal cancer. UpToDate. 2025. Accessed at https://www.uptodate.com/contents/epidemiology-and-risk-factors-for-esophageal-cancer on May 22, 2025.
Ku GY, Ilson DH. Chapter 71: Cancer of the Esophagus. In: Niederhuber JE, Armitage JO, Doroshow JH, Kastan MB, Tepper JE, eds. Abeloff’s Clinical Oncology. 6th ed. Philadelphia, Pa. Elsevier: 2020.
National Cancer Institute. Esophageal Cancer Treatment (PDQ®)–Patient Version. 2025. Accessed at https://www.cancer.gov/types/esophageal/patient/esophageal-treatment-pdq on May 22, 2025.
Saltzman JR, Gibson MK. Clinical manifestations, diagnosis, and staging of esophageal cancer. UpToDate. 2025. Accessed at https://www.uptodate.com/contents/clinical-manifestations-diagnosis-and-staging-of-esophageal-cancer on May 22, 2025.
Last Revised: August 14, 2025
American Cancer Society medical information is copyrighted material. For reprint requests, please see our Content Usage Policy.
This information is possible thanks to people like you.
We depend on donations to keep our cancer information available for the people who need it most.







