Your gift is 100% tax deductible.
How Is a Cytology Test Done?
Cytology testing is done to screen for and diagnose diseases, including cancer, by looking at cells under a microscope.
What is a cytology test?
Diagnosing diseases by looking at single cells and small clusters of cells under a microscope is called cytology or cytopathology testing. It’s an important part of diagnosing some types of cancer.
Cytology tests differ from biopsies because they only use a few cells, rather than a larger tissue sample. Compared with a biopsy, collecting a cytology sample is often:
- Easier to do
- Less likely to have serious complications
- Less painful
- Less costly
Compared with a biopsy, cytology tests may provide less detailed information. They cannot give information about how cells are arranged or how deeply a tumor might have grown into the tissue. However, they are often just as useful.
Cytology tests are used for two main purposes:
- Screening cytology tests can help find cancer before symptoms appear.
- Diagnostic cytology tests are used to test for disease when a person has signs or symptoms. They can also help evaluate abnormal screening results and often assist in classifying disease.
If cytology results suggest cancer, a biopsy is often done to confirm the diagnosis before treatment begins.
How are cytology samples collected?
Cytology samples can be collected in multiple ways to test for cancer:
With an FNA, a very thin, hollow needle attached to a syringe is used to remove (aspirate) a small amount of fluid and very small pieces of tissue from an abnormal area. This includes areas like lymph nodes, breast lumps, or thyroid nodules.
It is done when the area that is being sampled is near the skin and can be felt by the doctor. The needle is placed through the skin directly into the area to be sampled.
If the abnormal area is deeper inside the body, the needle can be guided into place using an imaging test, such as an ultrasound or CT scan.
This test may be considered a cytology test or a type of biopsy.
Various kinds of body fluids can be collected and tested using cytology to see if they contain cancer cells. Some of these include:
- Ascitic fluid (peritoneal fluid) from the belly
- Breast drainage
- Pericardial fluid from the sac that surrounds the heart
- Pleural fluid from the space around the lungs
- Spinal fluid (cerebrospinal fluid, CSF) from the space surrounding the brain and spinal cord
- Sputum (phlegm) from the lungs
- Urine (pee)
Cytology samples can also be collected by gently scraping or brushing cells from the organ or tissue being tested, sometimes called a brush biopsy. One example is the Pap test, which uses a small spatula or brush to collect cells from the lower part of the uterus (cervix).
Cells can also be collected from other areas by brushing or scraping, including the mouth and throat, esophagus, stomach, bile and pancreatic ducts, and the airways of the lungs. Sometimes fluid is used to collect washings (lavages) from areas such as the lung airways or digestive tract for cytology testing.
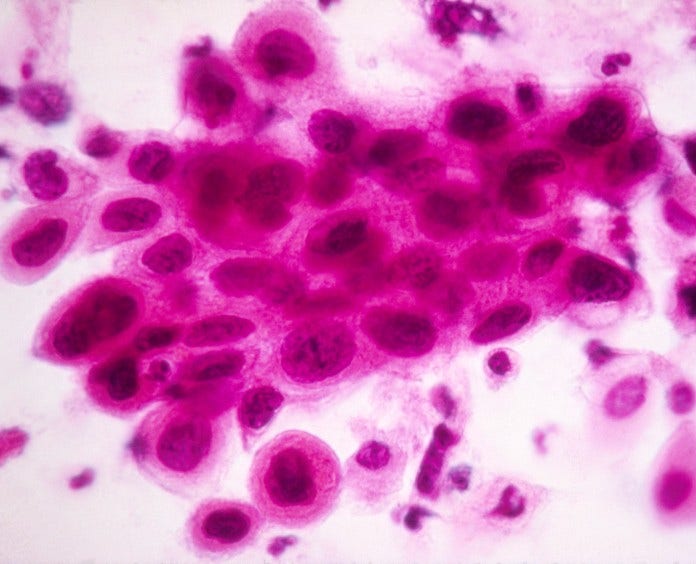
What a Pap test that shows cervical cancer looks like under the microscope
How long are cytology samples kept?
Processed cytology samples can be stored on slides or within paraffin (wax) blocks.
Why keeping samples can be helpful
Processed samples are usually kept for a set amount of time, which allows them to be sent to another doctor or lab for a second opinion. This process, called a cytopathology review, involves another doctor examining the sample to confirm or suggest a different diagnosis.
Keeping samples can also help if a new cancer occurs years later. Original and new slides can be compared to help determine if the cancer has come back (recurrence) or if it is new. Additional tests, like special stains on the original cells, might also be helpful.
Legal requirements
Clinical labs are regulated and certified based on a federal law called CLIA (Clinical Laboratory Improvement Amendments). To be CLIA accredited, labs must keep cytology samples for a minimum amount of time. For instance, CLIA says that labs must keep:
- Cytology slides for at least 5 years
- Paraffin blocks for at least 2 years
Some states have their own laws that require labs to keep cytology samples longer than the time specified by CLIA. And some labs have policies for keeping samples even longer than required by federal or state laws.
If you would like to know if the lab you go to is CLIA certified, you can ask the lab staff. Or if you have questions or concerns, you can reach out to your state’s CLIA agency.
- Written by
- References

Developed by the American Cancer Society medical and editorial content team with medical review and contribution by the American Society of Clinical Oncology (ASCO).
Centers for Medicare & Medicaid Services. Clinical laboratory improvement amendments (CLIA). Cms.org. Updated February 6, 2026. Accessed at https://www.cms.gov/medicare/quality/clinical-laboratory-improvement-amendments on March 5, 2026.
National Cancer Institute (NCI). SEER Training Module Bladder Cancer: Cytology Report. Accessed from https://training.seer.cancer.gov/bladder/cancer-evaluation/cytology-report.html on March 5, 2026.
Slaoui M, Bauchet A-L, Fiette L. Tissue sampling and processing for histopathology evaluation. Methods Mol Bio. 2017; 1641:101-114.
Tyagi S, Swasthi S. A textbook of cytology. Donimant: 2022.
Last Revised: March 24, 2026
American Cancer Society medical information is copyrighted material. For reprint requests, please see our Content Usage Policy.
American Cancer Society Emails
Sign up to stay up-to-date with news, valuable information, and ways to get involved with the American Cancer Society.



